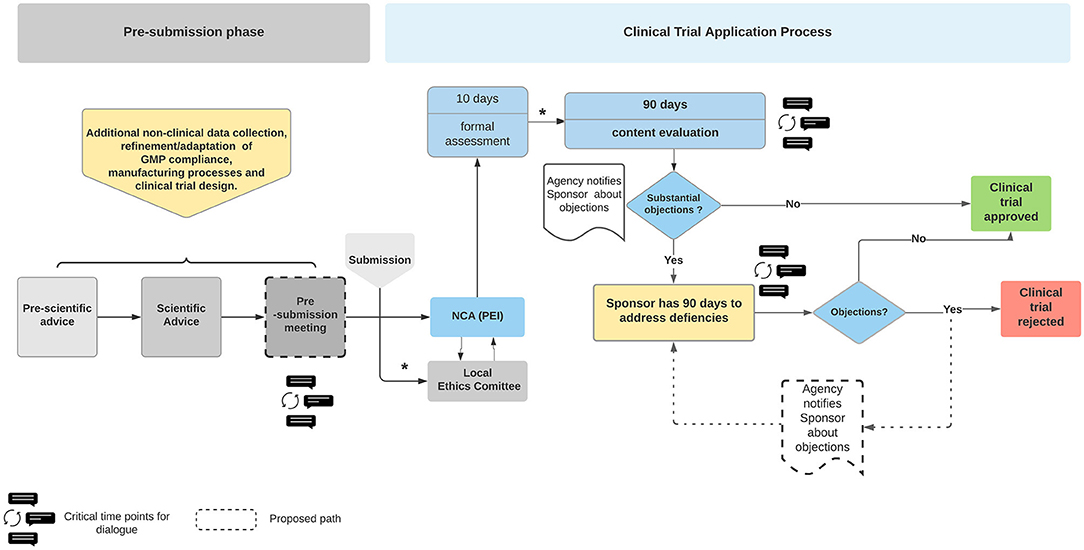
EMA & FDA Approvals and Recommendations in 2020 for Oncology Drugs and Diagnostics/Devices | CATO SMS
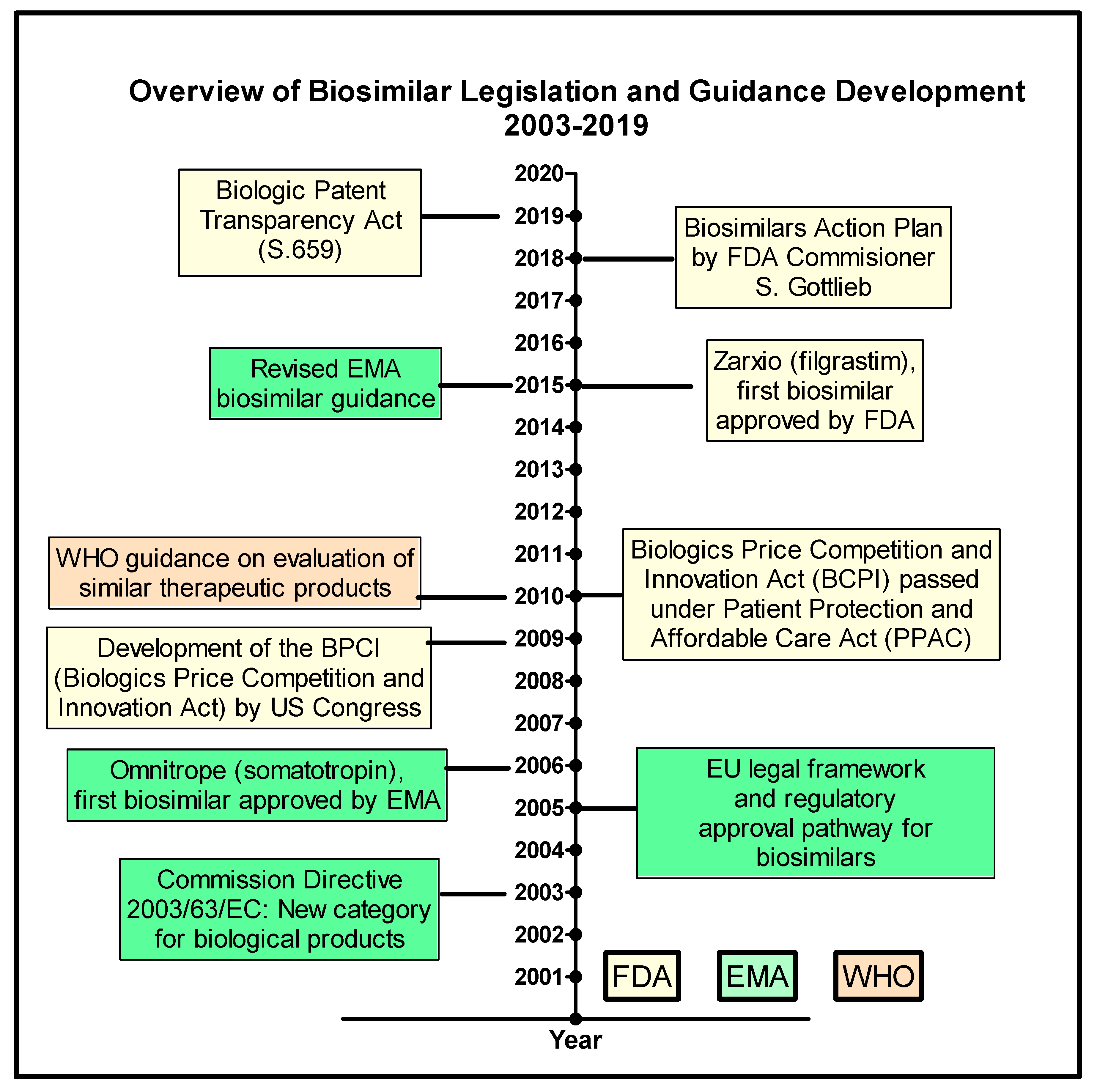
Pharmaceutics | Free Full-Text | The Biosimilar Landscape: An Overview of Regulatory Approvals by the EMA and FDA | HTML
Overview of comments - Requirements to the chemical and pharmaceutical quality documentation concerning investigational medicina

Comparison of regulatory pathways for the approval of advanced therapies in the European Union and the United States - Cytotherapy
Regulatory Affairs 101: Introduction to Investigational New Drug Applications and Clinical Trial Applications
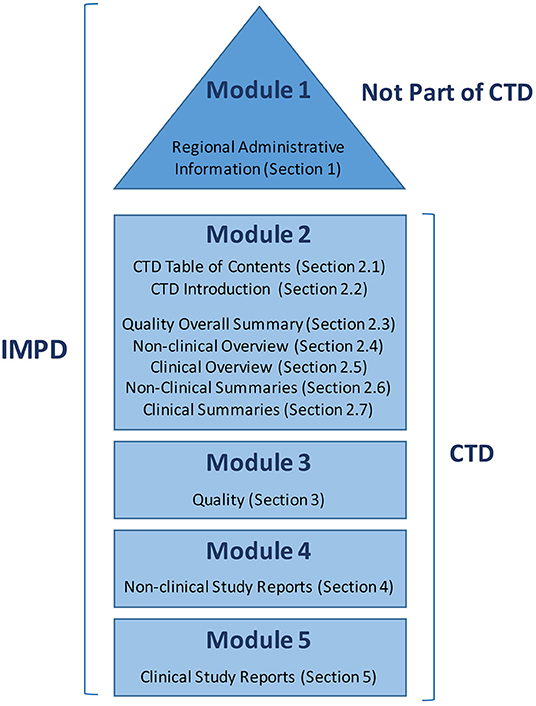
Frontiers | Transitioning From Preclinical Evidence to Advanced Therapy Medicinal Product: A Spanish Experience

Commentary on the EMA Guideline on strategies to identify and mitigate risks for first‐in‐human and early clinical trials with investigational medicinal products - Gerven - 2018 - British Journal of Clinical Pharmacology -
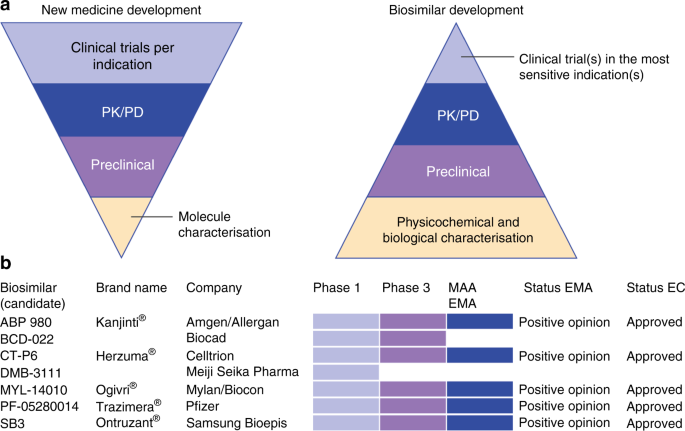
The arrival of biosimilar monoclonal antibodies in oncology: clinical studies for trastuzumab biosimilars | British Journal of Cancer
Reflection paper on guidance for laboratories that perform the analysis or evaluation of clinical trial samples