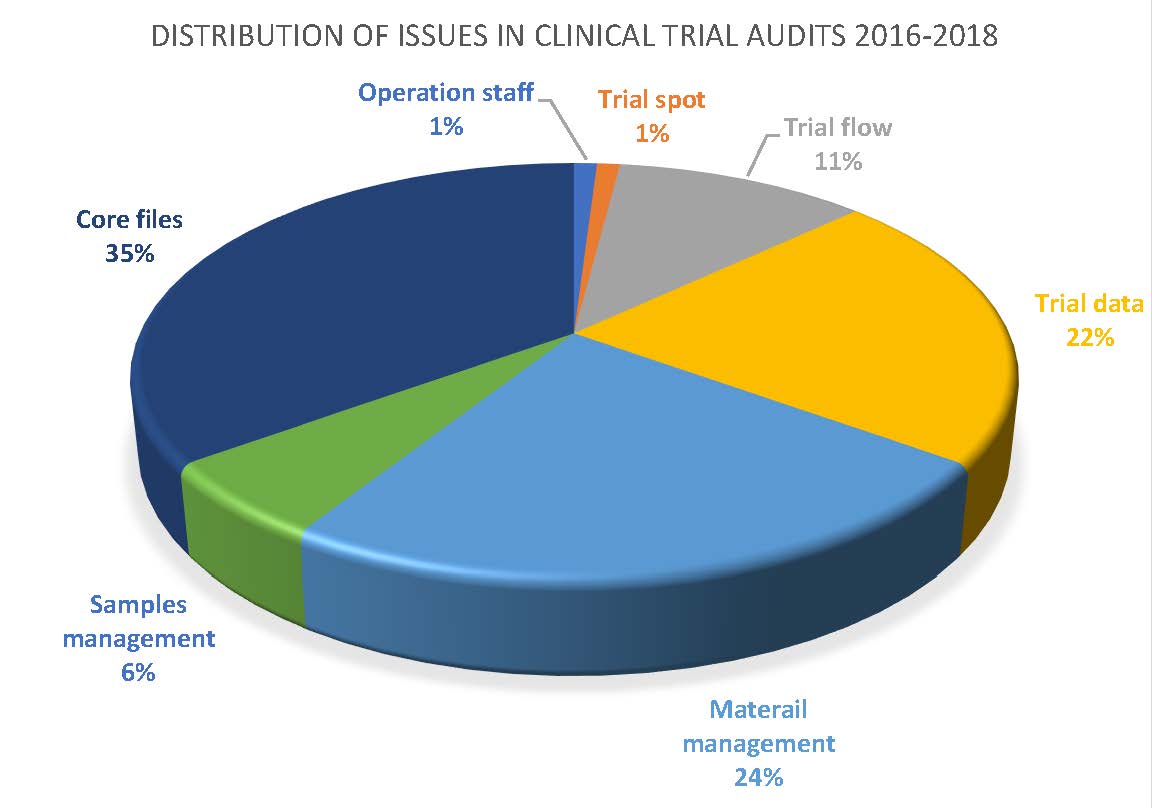
Potential Risks and Mitigation Strategies Before the Conduct of a Clinical Trial: An Industry Perspective | Bentham Science
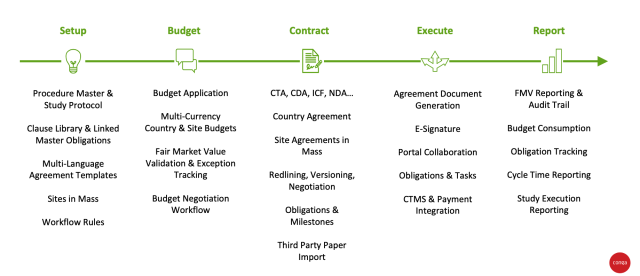
![PDF] Informed consent in clinical research: Consensus recommendations for reform identified by an expert interview panel | Semantic Scholar PDF] Informed consent in clinical research: Consensus recommendations for reform identified by an expert interview panel | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/3f6b229a2937974f4f37094037fcb258b6a8d47d/3-Figure1-1.png)
PDF] Informed consent in clinical research: Consensus recommendations for reform identified by an expert interview panel | Semantic Scholar
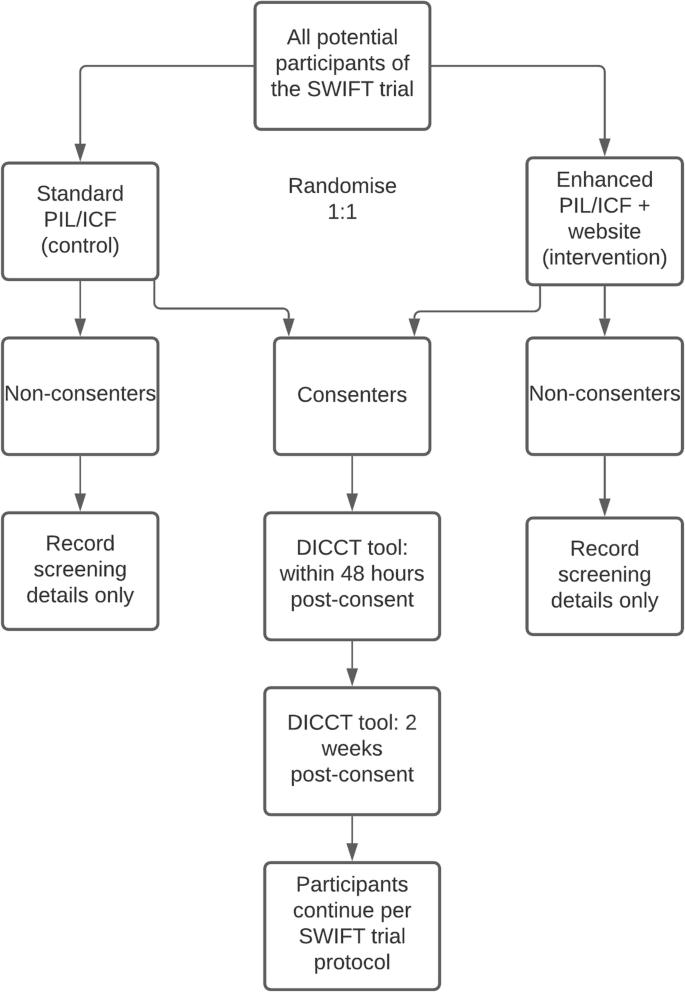
An enhanced participant information leaflet and multimedia intervention to improve the quality of informed consent to a randomised clinical trial enrolling people living with HIV and obesity: a protocol for a Study